Effect of indigenous extraction solvents on the phytochemical, GC-MS profile and antioxidant activities of Ocimum gratissimum extracts
Keywords:
Phytochemicals, Extraction solven, GC-MS profiling, Ocimum gratissimumAbstract
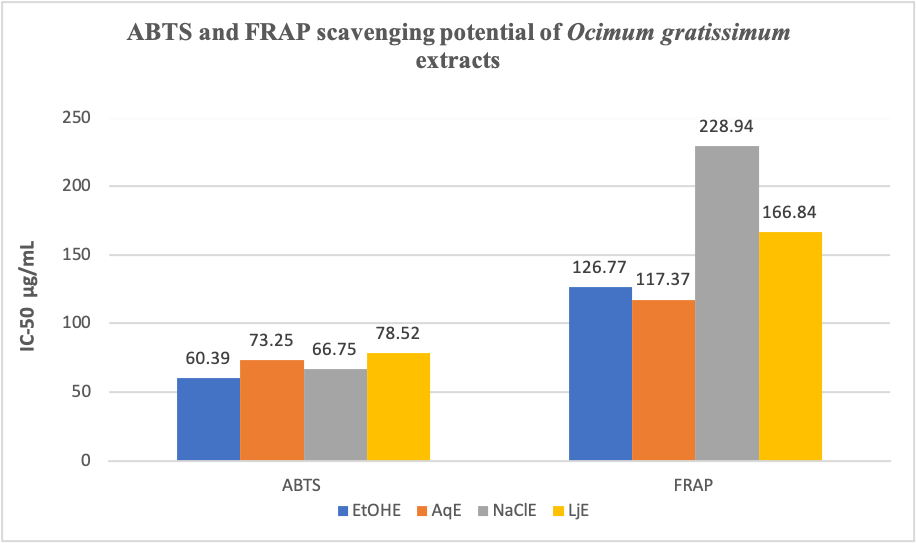
This study compared the effects of Ethanol and indigenous extractants (Aqueous, 2% NaCl solution, and 40% v/v Lime juice) on the bioactive properties of Ocimum gratissimum extract. Phytochemical, GC-MS, and antioxidant analysis of the Ethanol (EtOHE), Aqueous (AqE), NaCl solution (NaClE), and Lime juice (LjE) extracts were carried out. All the extracts contained saponins, tannins, flavonoids, phenols, steroids, and alkaloids, with Ethanol and Aqueous extracts showing higher concentrations of these compounds compared with NaCl and Lime juice extracts (p < 0.05). GC-MS characterization identified twenty-one bioactive compounds in the Ethanol extract, while only fifteen compounds were found in the Aqueous, NaCl, and Lime juice extracts, respectively. The Ethanol extract had higher Total Antioxidant Capacity (TAC) (26.08 ± 0.76 μg/ml) compared with Aqueous (7.79 ± 0.61 μg/ml), NaCl (7.82 ± 0.81 μg/ml), and Lime juice (3.49 ± 0.48 μg/ml) extracts (p < 0.05). The Aqueous extract had the strongest Ferric reducing antioxidant power, with a lower IC50 value of 117.37 μg/ml, compared with NaCl (228.94 μg/ml) and Lime juice (166.84 μg/ml) extracts, while Ethanol extracts had the lowest IC50 value (60.39 μg/ml) for ABTS radicals compared with NaCl (66.75 μg/ml) and Lime juice (78.52 μg/ml) extracts. In conclusion, all the extracts exhibited potential medicinal and antioxidant properties; however, the bioactive and antioxidant properties of the extracts demonstrate a dependence on the extraction methods and solvents. It is therefore important to always ascertain which extraction solvent and method is appropriate for the preparation of safe medicinal herbs.
Published
How to Cite
Issue
Section
Copyright (c) 2026 Tunmise T. Oladipe, Ebenezer O. Oladipe, Abayomi A. Taiwo, Ademuyiwa J. Adegbegi, Ajayi Ajetomobi, David A. Ibiyemi, Ekundayo T. Areh

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- M. A. Aliu, A. M. Junaid, A. Ibraheem, A. Ishaq, A. Lawal, K. E. Ayeni, A. R. Lawal, L. B. Abdulrauf, Dispersive liquid-liquid microextraction/HPLC techniques for determination of oxytetracycline and doxycycline residues in beef samples: method developments and statistical analysis , African Scientific Reports: Volume 2, Issue 1, April 2023
- E. Agboeze, N. P. Ani, E. O. Omeje, Extraction and Characterization of Pharmaceutical Grade Microcrystalline Cellulose From Bambara Nut (Voandzeia Subterranean (L) Thousars) Husk , African Scientific Reports: Volume 1, Issue 2, August 2022
- Oluwagbenga Anifowose, Bitrus W. Tukura, Obaje D. Opaluwa, Detoxification of Ochratoxin A in Rice and Maize using ethanolic leaf extracts of Moringa oleifera and Vernonia amygdalina , African Scientific Reports: Volume 4, Issue 2, August 2025
- O. U. Igwe, C. C. Oru, I. E. Otuokere, Chemical and bioprotective studies of Xylopia aethiopica seed extract and molecular docking of doconexent and cryptopinone as the prominent compounds , African Scientific Reports: Volume 3, Issue 2, August 2024
- Ifeanyi Edozie Otuokere, Onyinye Uloma Akoh, Felix Chigozie Nwadire, Chinedum Ifeanyi Nwankwo, Joy Nwachukwu Egbucha, Chiemela Wisdom, Ogbonna Augustine Okwudiri, GC-MS Profiling and In Silico Studies to Identify Potential SARS-CoV-2 Nonstructural Protein Inhibitors from Psidium guajava , African Scientific Reports: Volume 1, Issue 3, December 2022
- Matthew Olaleke Aremu, Stephen Olaide Aremu, Ibrahim Aliyu, David Bala Passali, Munir Hussaini, Benjamin Zobada Musa, Rasaq Bolakale Salau, Amino acids profile and health attributes of Bambara groundnut (Vigna subteranea L.) and sesame (Sesamum indicum L.): a comparative study , African Scientific Reports: Volume 4, Issue 1, April 2025
- Adenike Adegoke-Elijah, Theresa Omolayo Ojewumi, Kudirat Oyewumi Jimoh, ECG anomaly detection: a deep learning perspective with LSTM encoders , African Scientific Reports: Volume 4, Issue 3, December 2025
- E. K. Ukpoko, I. S. Eneji, Q. M. Amua, R. A. Wuana, Analysis of some heavy metals in foodstuffs contaminated with pesticides using a developed spot-test method , African Scientific Reports: Volume 4, Issue 1, April 2025
- Nwamaka I. Akpu, K. U. P. Okpechi, Elizabeth C. Nwaokorongwu, Imosobomeh L. Ikhioya, Julian C. Onwuzo, Lebe A. Nnanna, Ifeanyi I. C Agbodike, Impact of temperature difference on the features of spray deposited yttrium doped cobalt selenide (YCoSe) thin films for photovoltaic application , African Scientific Reports: Volume 2, Issue 3, December 2023
- D. Raymond, T. P. Pantuvo, A. Lydia, J. Sabo, R. Ajia, An Optimized Half-Step Scheme Third derivative Methods for Testing Higher Order Initial Value Problems , African Scientific Reports: Volume 2, Issue 1, April 2023
You may also start an advanced similarity search for this article.




