Numerical evaluation of energy shifts due to dipole moments interaction in a hydrogen atom
Keywords:
Numerical methods, Energy shifts, Hydrogen atom, Wave function, Dipole-dipole interaction and Stark effectAbstract
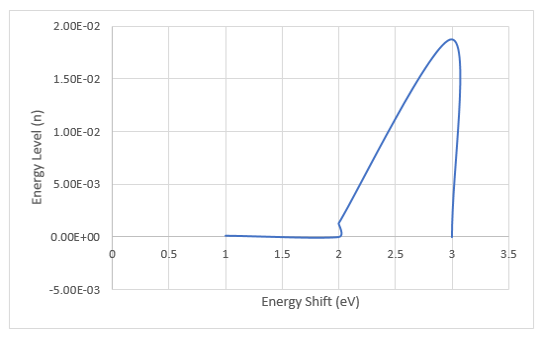
This study aimed to numerically investigate the impact of dipole-dipole interactions on the energy levels of a hydrogen atom, specifically the energy shift caused by the interaction of the atom's dipole moment with external electric fields. This research involved applying perturbation theory and numerical techniques to quantify the changes in energy levels due to dipole orientations and magnitudes, contributing to a deeper understanding of atomic behavior and its implications in various scientific disciplines. The results presented showed that the wavefunction of the hydrogen atom exhibits degeneracy, especially n=2, n=3 states. This implies that n=2, n=3, have different states but the same energy as a result of the subshells with angular momentum l, which contains 2l+1 distinct state corresponding to positive value of m (magnetic angular momentum quantum number) m=-l±1. The total degeneracy of the energy levels n is caused by n2. We find out that as the orientation of the electron charges (θ,ϕ) change the dipole moment also changes and thus has a perturbing effect on the energy levels of the hydrogen atom leading to a shift in its energy. This is confirmed, as the energy shift increases slightly from the ground state energy function Φ100 to the second exicted state Φ300, while the degeneracy state of Φ310 experiences infinitesimal energy shift above. This is because the probability of the electron been found in the lower energy level is greater than at higher energy level and so it is mostly affected there by energy shift. Also, the dipole moment could increase liberational energy of the atom and thus lead to more excitement even though its impact is quite negative. The outcome of this study could have implications in precision measurements, quantum computing, and the understanding of complex atomic systems.
Published
How to Cite
Issue
Section
Copyright (c) 2026 Otor Daniel Abi, Anene Gerald Makuachukwu, Daniel Ominyi Sunday, Peter Ikpe Adoga, Ortwer Felix Igbasue

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Louis Tersoo Abiem, Tertsea Igbawua, Jacob Tersugh Adawa, Investigation of direct normal irradiance and total cloud cover connection over Nigeria using satellite data , African Scientific Reports: Volume 4, Issue 3, December 2025
- Abayomi Ajayi, Olaniyi Olayiwola, Fadeke Apantaku, Idowu Osinuga, Oluwaseun Wale-Orojo, Predicting the needs of people living with a disability using the two-level logit-skewed exponential power model , African Scientific Reports: Volume 3, Issue 2, August 2024
- Gafari Abiola Adepoju, Muniru Olajide Okelola, Muhammed Adekilekun Tijani, Hybrid Optimization Technique for Solving Economic Dispatch Problem: A Case Study of Nigerian Thermal Power System , African Scientific Reports: Volume 1, Issue 2, August 2022
- Shadreck Muyambo, Jack A. Urombo, Temperature-dependent viscometry of baobab pectin (Adansonia digitata L.) , African Scientific Reports: Volume 3, Issue 1, April 2024
- Rwat Solomon Isa, Tsok Samuel Hwere, Usman Garba, Shehu Sidi Abubakar, Sabastine Emmanuel, Mathematical modelling of terrorists re-cycle induced with backward bifurcation , African Scientific Reports: Volume 2, Issue 3, December 2023
- E. U. Chukwuegbo, E. Agboeze, C. C. Ogbuanu, S. Amujiogu, Effect of Phthalonitrile and Alum on Flame Retardation Properties of 100% Cotton Fabric , African Scientific Reports: Volume 1, Issue 2, August 2022
- Ebenezer O. Ayoola, Victor N. Enwemiwe, Hilda O. Erhenhi, Clement C. Obi, Agboola A. Adebowale, Eric Esiwo, Joyce A. Onokpasa, Robert B. Ikomi, Acute toxicity of Oreochromis niloticus fingerlings exposed to effluent from Delta State University, Abraka Nigeria boys hostel , African Scientific Reports: Volume 3, Issue 2, August 2024
- Peace P. Desben, Emmanuel N. Dashe, Joel N. Ndam, Transmission dynamics of community-and hospital-acquired infections: insights from mathematical modelling , African Scientific Reports: Volume 5, Issue 2, August 2026
- Taiwo J. Adejumo, Kayode Ayinde, Abayomi A. Akomolafe, Olusola S. Makinde, Adegoke S. Ajiboye, Robust-M new two-parameter estimator for linear regression models: Simulations and applications , African Scientific Reports: Volume 2, Issue 3, December 2023
- D. Raymond, T. P. Pantuvo, A. Lydia, J. Sabo, R. Ajia, An Optimized Half-Step Scheme Third derivative Methods for Testing Higher Order Initial Value Problems , African Scientific Reports: Volume 2, Issue 1, April 2023
You may also start an advanced similarity search for this article.




