Dispersive liquid-liquid microextraction/HPLC techniques for determination of oxytetracycline and doxycycline residues in beef samples: method developments and statistical analysis
Keywords:
beef, liquid chromatography, microextraction, tetracyclines, validation`Abstract
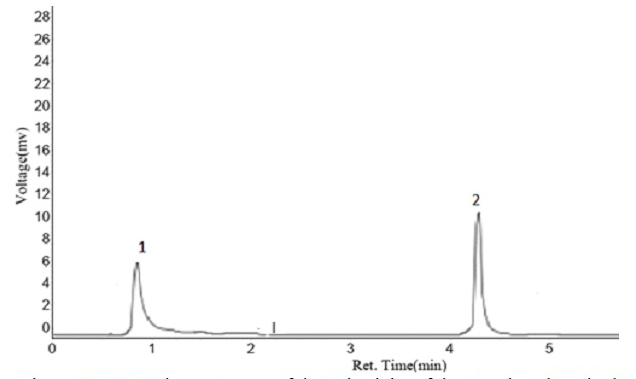
A rapid, cost-effective and environment-friendly sample pre-treatment method involving dispersive liquid-liquid microextraction (DLLME) and high-performance liquid chromatography (HPLC) was developed and applied for the extraction of oxytetracycline and doxycycline residues in beef samples (liver, kidney and muscle). Several influencing factors associated with the extraction and separation of these antibiotics residues, such as sample size, type and volume of disperser and extraction solvents, centrifugation speed and time, were optimized using Plackett-Burman design and central composite design, while insignificant factors were fixed at values determined using univariate analysis. Figures of merit of the analytical methodology including the limit of detection (LOD), the limit of quantification (LOQ), accuracy (in terms of average recoveries), precision and calibration functions were established according to the European Union commission decision 2002/657/EC. Linearity, in the range of 5–500 µg/kg, was obtained with regression coefficients ranging from 0.9983 – 0.9999. Inter-day repeatability, intra-day precision, LODs and LOQs obtained were 3.81 – 14.90%, 3.80 – 8.70%, 4.21 – 4.69 µg/kg and 14.02 – 15.65 µg/kg respectively. Samples with detectable drug residues have oxytetracycline being the most commonly detected. The developed method was successfully established and the concentration levels of drug residues detected were lower than the European Union set maximum residue level (MRL).
Published
How to Cite
Issue
Section
Copyright (c) 2023 M. A. Aliu, A. M. Junaid, A. Ibraheem, A. Ishaq, A. Lawal, K. E. Ayeni, A. R. Lawal, L. B. Abdulrauf

This work is licensed under a Creative Commons Attribution 4.0 International License.



