Natural Skin Whitening Compounds: Types, Source and Mechanism of Action: A Review
Keywords:
Skin whitening, Bleaching, Melanin, MelanogenesisAbstract
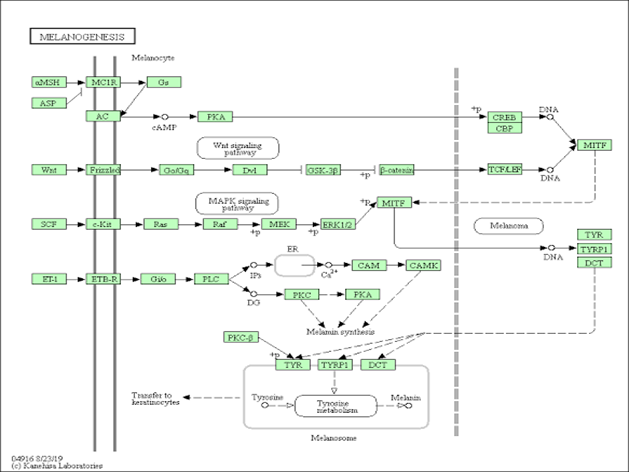
Skin whitening, also known as skin lightening or skin bleaching, is utilizing chemical compounds to lighten or even out the skin’s colour by lowering the skin’s melanin concentration. The biosynthetic process of melanogenesis and the related main regulatory signaling pathways are summarized in this paper. It also addresses the efficacy of natural skin-whitening treatments based on their mode of action on melanogenesis and their compound classification. The review’s goal is to provide useful information and create awareness on the effects of bleaching cream.
Published
How to Cite
Issue
Section
Copyright (c) 2023 Chiagoziem Wisdom Chidiebere, Ugochukwu Aniefuna Azuka Anthonia

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Similar Articles
- Solomon Denen Igba, Idugba Mathias Echi, Emmanuel Vezua Tikyaa, Simulation of thermochemical effects on unsteady magneto hydrodynamics fluids flow in two dimensional nonlinear permeable media , African Scientific Reports: Volume 3, Issue 3, December 2024
You may also start an advanced similarity search for this article.




